UCSF Pediatrics Professor Jennifer Puck, MD, has spent decades pushing for universal newborn screening to detect severe combined immune deficiency (SCID), a rare but deadly genetic disorder. Today, her grit was affirmed by a Lancet study directly showing that, by enabling earlier treatment, newborn screening was the most significant factor in improving survival outcomes in children with SCID in the last 40 years.
Children with SCID, or bubble baby disease, are born without a functioning immune system, so that even a common cold can be life-threatening. Identifying SCID early is the key to treatment early in life, before onset of infections.
Co-led by UCSF transplant specialist Christopher Dvorak, MD and colleagues with the National Institutes of Health-funded Primary Immune Deficiency Treatment Consortium (PIDTC), the Lancet study reviewed more than 900 cases of children diagnosed with SCID between 1982 and 2018. The researchers found that SCID newborn screening – and the early treatment it made possible – raised the five-year survival rate to 87% in the time period from 2010 to 2018, when the screening was increasingly being used. In the subset of babies who received newborn screenings, the survival rate at five years was 92.5%. That’s up from 73% survival before newborn SCID screening was invented or common (1982 to 2009).
“It surprised us!” said Puck, who directs the UCSF Jeffrey Modell Diagnostic Center for Primary Immunodeficiencies. “Despite all our advances – better antibiotics, better ways to do transplants – there really was no improvement in survival until newborn screening showed up. We knew delivering treatment right away made a difference in survival; we just didn’t expect it to be the only factor to account for the difference.”
Despite all our advances – better antibiotics, better ways to do transplants – there really was no improvement in survival until newborn screening showed up.
Jennifer Puck, MDUCSF Jeffrey Modell Diagnostic Center for Primary Immunodeficiencies
This is not to say that transplants and new drugs aren’t important, Puck was quick to add – they improve other outcomes such as comorbidities and quality of life, and future studies will continue to examine those. Treatments that Puck, Dvorak and other UCSF colleagues have worked on, such as autologous gene therapy, for example, have shown remarkable success in helping kids with the very deadliest forms of the disease.
But when it comes to survival, timing is almost everything.
From the lab to the legislature
Puck herself conceived of and developed the SCID newborn screening test in 2005 while working as a scientist at the National Human Genome Research Institute at the National Institutes of Health. The test works by detecting a lack of infection-fighting T cells in the dried bloodspots that have long been collected from newborns’ heels for various screenings.
After moving to California in 2006, Puck helped convince California lawmakers to require the screening statewide, which they eventually did in 2010, becoming the third state to do so.
“I’d go to legislative hearings with families,” Puck recalled. “One mother described losing her first child to SCID while she was pregnant with her second, and there wasn’t a dry eye in the room. Meanwhile, that second child – whose SCID was detected early – was running all around with his big blond curls, looking like a million bucks.
“Sure enough, the committee unanimously approved the screening,” Puck added. “Why should a family have to endure the tragedy of losing one baby with SCID before being able to recognize and save the second with early treatment? A population-wide newborn screening test identifies SCID at birth.”
She continued her advocacy and, eventually, all 50 states adopted SCID screening as part of their standard newborn panel, with the last three doing so in 2018.
“California has an eighth of all babies born in the U.S., and half a million babies born a year, so getting this state on board provided a big impetus for other states to follow,” said Puck.
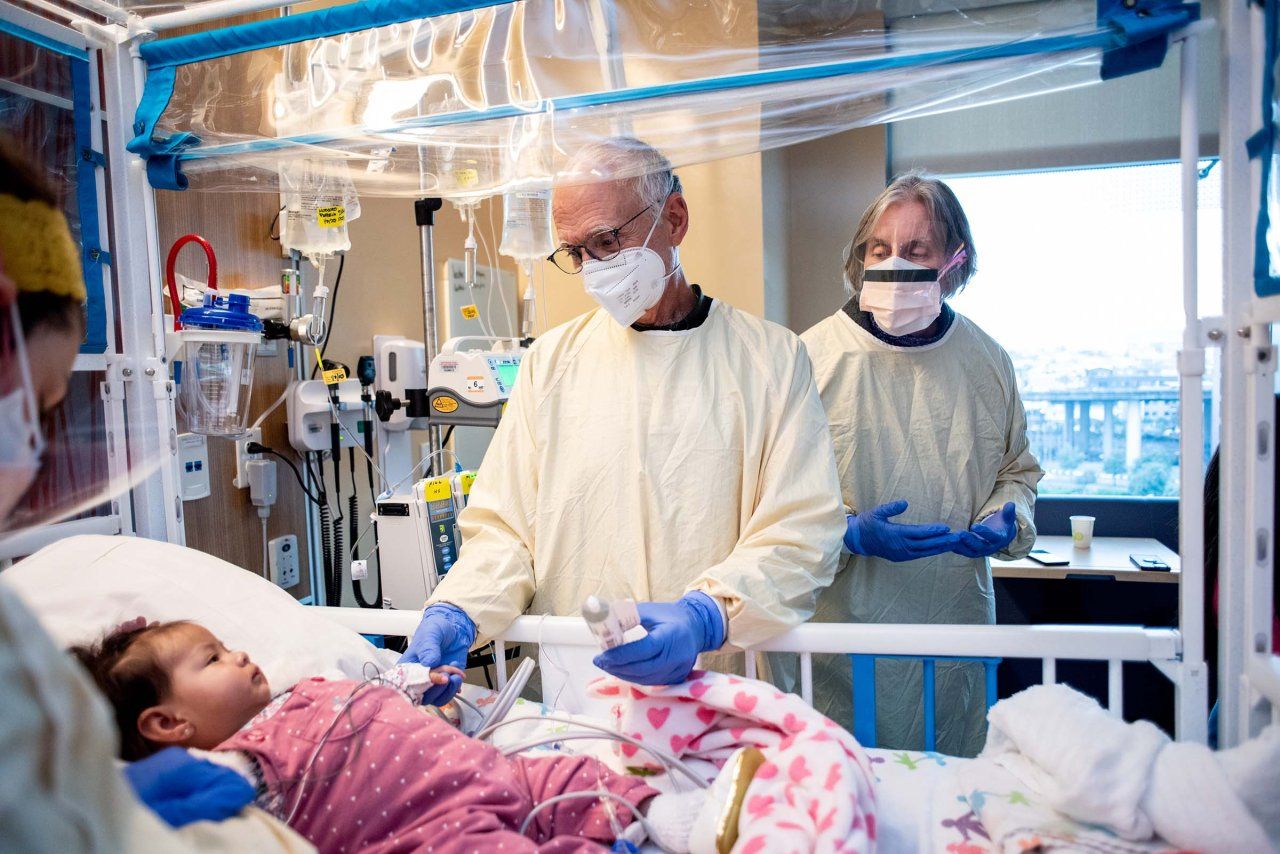
Moving from an emergency to a plan of care
SCID is typically fatal within the first two years of life, unless the immune system is restored through stem-cell transplant, gene therapy or enzyme therapy.
Before newborn screening was in place in California, SCID patients often arrived at UCSF Benioff Children’s Hospitals very ill, said Dvorak, who is chief of the UCSF Pediatric Allergy, Immunology and Bone Marrow Transplantation division.
“The SCID screening is the only test or process I can think of that has completely and utterly shifted how we care for these patients from beginning to end,” said Dvorak. “It has gone from being this huge emergency – like, we need to transplant this kid right now because they are dying from a horrible infection – to having a kid who looks good, has been properly isolated, hasn’t had an infection, and whose family has time to prepare for the transplant.”

Prior research has shown that being younger than 3.5 months at the time of bone marrow transplant – the main treatment for SCID – and not having an active infection improves five-year survival rates for children with SCID. Both of those factors are facilitated by newborn screening.
Push to expand screening worldwide
Puck and Dvorak hope the new Lancet study – which was funded by the National Institutes of Health – will persuade countries outside the U.S. to adopt newborn screening for SCID too. Some have resisted because of money, logistics and concern about making parents anxious, they said.
“The screening isn’t just a test, but a program that requires a lot of work and quality control – doing the test, contacting the family, a plan for when the test is positive,” Puck said. “One objection is that false positives can really upset a lot of parents.”
However, the SCID test has the lowest rate of false positives and false negatives of any newborn screening test in existence, she added.
“With some other tests, a positive result will end up with only 1% of those babies actually having the condition,” Puck said. “For the SCID test, almost 50% of those with a positive test have, at the least, low T cells. Even if they don’t have SCID, they should be seen by immunology experts.”
Research by UCSF colleague Lena Winestone, MD, an assistant professor of pediatrics, suggests another impressive aspect of universal SCID newborn screening: it may eliminate racial disparities in treatment access and outcomes for children with the disorder.
“The SCID newborn screening test is a leveler,” said Puck. “It’s about fairness and providing screening and care so that all kids, regardless of race or background, can have their healthiest, longest life.”
