The ideal therapy for a disease works exclusively at the site of the disease.
But when it comes to the brain, which is wrapped in a protective barrier and contains thousands of different cell types, this ideal is very hard to achieve.
Now, scientists at UC San Francisco have been awarded more than $30 million by the Advanced Research Projects Agency for Health (ARPA-H) to lead the Cell Therapies for Neuroinflammation and Neurodegeneration (CT-NEURO) project, which will develop a new technology dubbed “tissue GPS.” The system uses engineered T cells to guide therapies directly to their targets in the brain to treat neurological diseases, like the deadly brain cancer glioblastoma, multiple sclerosis and Alzheimer’s.
These immune cells navigate by detecting molecular markers called antigens that appear on the surface of target cells like street addresses, replete with nine-digit zip codes. They only transfer their payload of anti-inflammatories or cancer-killing molecules once they have arrived at the right location.
The tissue GPS system may give immune cells the ability to pass through molecular gates in the blood brain barrier; find and stick to target cells; and safely deliver a tailored therapy. With such an exacting approach, the hope is that patients will be spared many of the uncomfortable or even dangerous side effects associated with other therapies.
“The field has many molecular tools that could potentially address conditions like neuroinflammation or Alzheimers, but it is challenging to know how to use these tools in a way that would precisely target the brain,” said Wendell Lim, PhD, project lead, UCSF Byers Distinguished Professor of Cellular and Molecular Pharmacology, and director of the UCSF Cell Design Institute. “Tissue GPS will ensure that therapies have a maximal effect in the right parts of the brain, making it much easier to treat complex disease.”
Disbursed over five years, the funds will support Lim and UCSF colleagues Scott Zamvil, MD, PhD; Hideho Okada, MD, PhD; Dean Sheppard, MD; and Anna Molofsky, MD, PhD, in developing the system.
The UCSF researchers will test the system’s delivery of therapies for brain tumors; neuroinflammation; demyelination (the loss of insulation on neural wires, like in multiple sclerosis); and neurodegeneration. They also will test its ability to reach other specific organs, like the lungs.
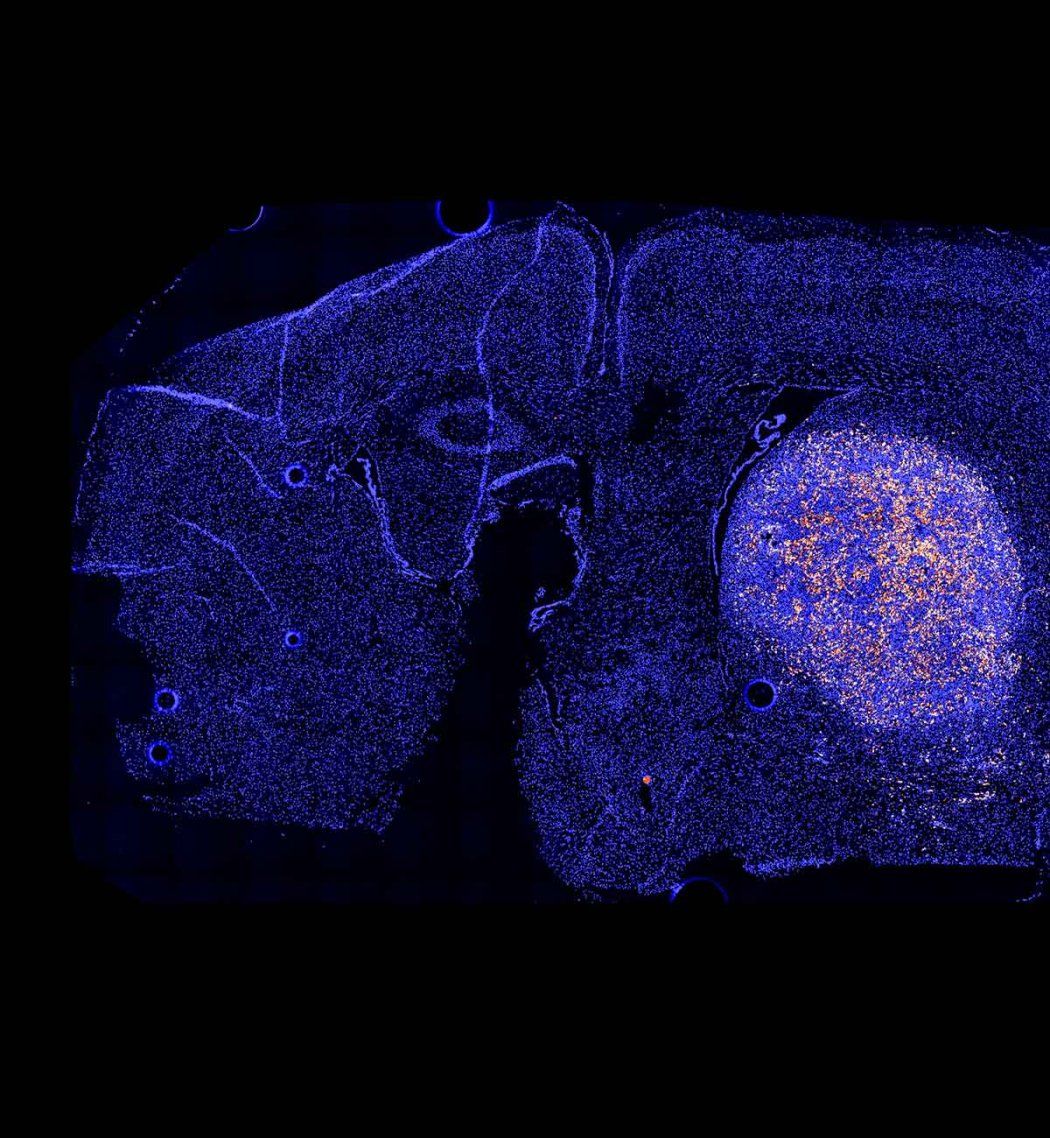
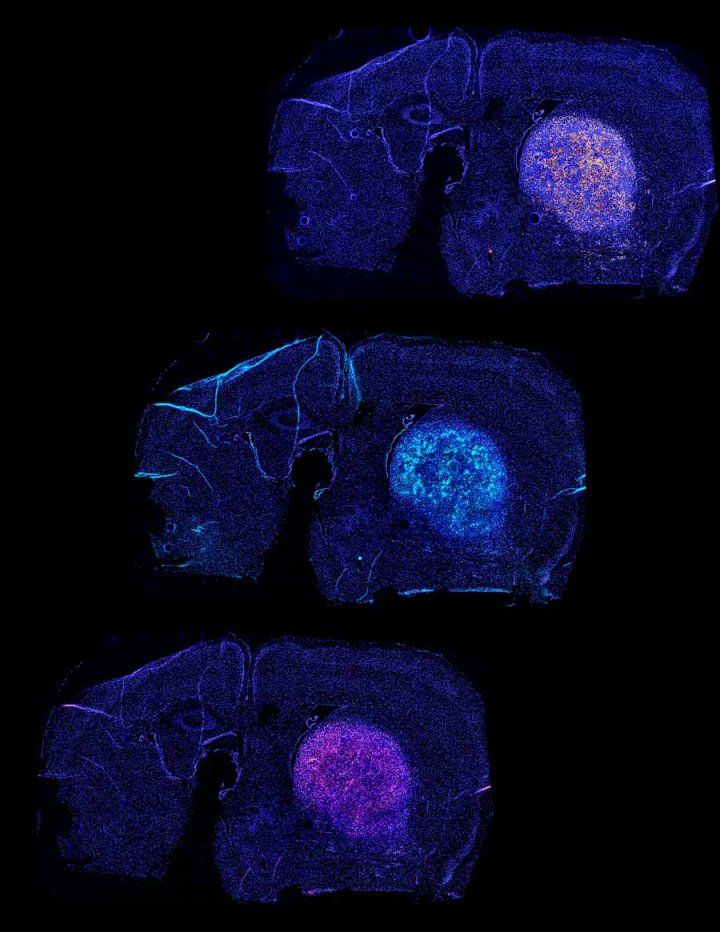
The team has mapped out antigens that are present throughout the brain, as well as the molecular sensors required for the tissue GPS to navigate to various brain regions. In an early experiment, their approach enabled engineered T-cells to treat brain tumors in mice.
“Immune cells are incredibly adept at traversing the body to reach a target,” Lim said. “These living cells could provide a powerful way to get therapies to where they need to be.”