A type of gene therapy called CAR T that has extended survival for thousands of patients with leukemia and other blood cancers is being adapted at UC San Francisco to treat people with glioblastoma, the most common and deadly adult brain tumor.
This new more powerful version of CAR T employs a novel technology developed at UCSF called synthetic notch (synNotch) that both protects healthy tissue from damage and enables the treatment to work more effectively.
UCSF opened enrollment this week for a clinical trial that is using the technology for the first time in people. A second trial, also at UCSF, is slated for 2025.
Approximately 12,000 Americans are diagnosed each year with glioblastoma. Patients survive on average for just 15 months after their diagnosis, and new treatments are urgently needed.
“This project is a prime example of bench-to-bed translation within UCSF, representing the strengths in basic and clinical science,” said Hideho Okada, MD, PhD, a physician-scientist and director of the UCSF Brain Tumor Immunotherapy Center. “We have a truly home-grown project here.”
Okada has received up to $11 million for the first trial from the California Institute for Regenerative Medicine (CIRM), which funds stem cell and gene therapy research for incurable diseases and disorders.
“We hope that the treatment will prolong lives for patients with glioblastoma,” said Okada, who is a professor of neurosurgery at UCSF and a member of the UCSF Weill Institute for Neurosciences. “However, the primary goal of the current phase 1 study is to ensure safety and characterize any toxicities.”
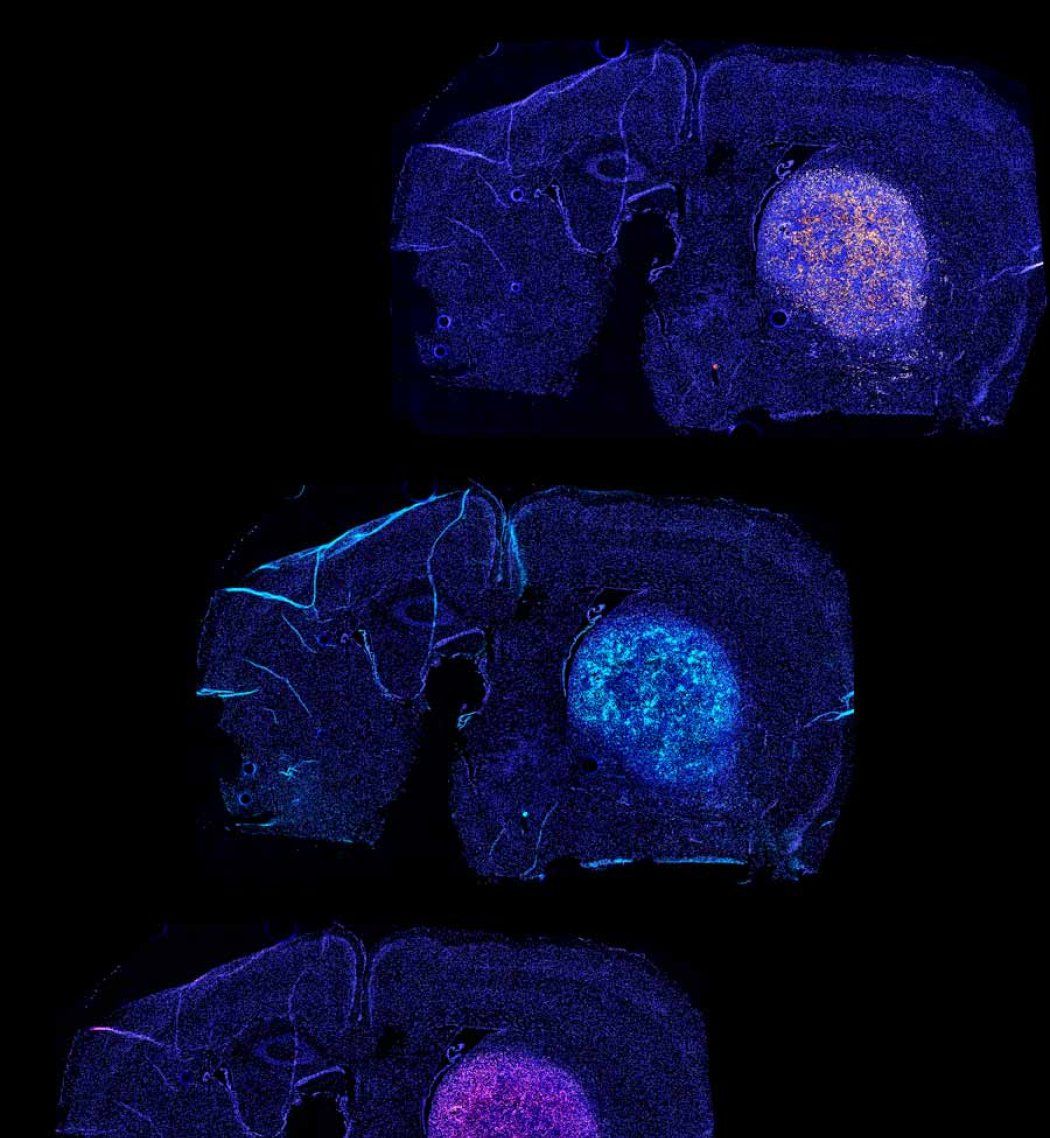
When tested in mice, Okada said the therapy provided a “robust and long-lasting result” that was more remarkable than anything he had encountered during 30 years of brain tumor research.
A lasting therapy that is designed to reduce side effects
The CIRM-funded trial will be led by principal investigator Jennifer Clarke, MD, MPH. It is open to patients with newly diagnosed glioblastoma who have completed standard-of-care treatment. Tumors must have a mutation found in approximately 20% of glioblastomas, and that can be identified by the UCSF500 cancer gene panel test.
The second study will be open to glioblastoma patients whether or not they have the mutation.
CAR T refers to chimeric antigen receptor T-cells, which are cancer-killing immune cells that have been extracted from the patient and genetically modified to recognize and destroy antigens that appear on the surface of cancer cells. These supercharged CAR T cells are then infused back into the body to attack tumor cells.
For many patients with leukemia and other blood cancers, CAR T has demonstrated long-term remission, but the approach hasn’t worked against brain tumors. Glioblastoma cells are more diverse than blood cancer cells, and they can evade CAR T. Many of the antigens made by the tumors are also found in healthy tissue, leaving them open to attack.
To overcome these obstacles, Okada drew from the synNotch system developed by Wendell Lim, PhD, director of the UCSF Cell Design Institute and professor in the UCSF Department of Cellular and Molecular Pharmacology.
The technology allowed scientists to program CAR-T cells to target specific antigens on tumor cells, without touching those found in healthy tissue. They also do not succumb to T-cell exhaustion, a common problem with CAR-T therapies, because they are more metabolically stable and use less energy to fight cancer longer.
“We’ve created a system that is flexible and thorough and addresses the major concerns we’ve had about using CAR-T cells against solid tumors,” Lim said. “These cells act like computers: integrating multiple units of information and making complex decisions.”
For the initial phase of development, Okada and colleagues were funded by the Parker Institute for Cancer Immunotherapy (PICI), UCSF Living Therapeutics Initiative (LTI), Sandler Foundation’s Glioma Precision Medicine Program (GPMP) and Friends of Michael Tilson Thomas.
Okada and his team recently received the prestigious National Cancer Institute’s Specialized Programs of Research Excellence (NCI SPORE) award to develop the second version of synNotch CAR T, which will be used for a wider patient population with glioblastoma. The second version has received additional support from the Mary Jane and Carl Panattoni Charitable Foundation, PICI, LTI and GPMP.
Editor’s note: this story was updated Aug. 16, 2024, with additional funding information.
About the California Institute for Regenerative Medicine (CIRM): At CIRM, we never forget that we were created by the people of California to accelerate stem cell treatments to patients with unmet medical needs, and act with a sense of urgency to succeed in that mission. To meet this challenge, our team of highly trained and experienced professionals actively partners with both academia and industry in a hands-on, entrepreneurial environment to fast track the development of today’s most promising stem cell technologies. With $5.5 billion in funding and more than 150 active stem cell programs in our portfolio, CIRM is one of the world’s largest institutions dedicated to helping people by bringing the future of cellular medicine closer to reality.