A small molecule called ISRIB that was identified at UC San Francisco can reverse the neuronal and cognitive effects of concussion in mice weeks after an injury occurred, new research has found.
ISRIB blocks the integrated stress response (ISR), a quality control process for protein production that can disrupt the normal function of cells when it is chronically activated.
The study, publishing Oct. 10, 2022, in Proceedings of the National Academy of Sciences, found that ISRIB reverses the effects of traumatic brain injury (TBI) on areas of neurons called dendritic spines that are critical to cognition. Mice treated with the drug also showed sustained improvements in working memory.
“Our goal was to see if ISRIB could ameliorate the neural effects of concussion,” said Michael Stryker, PhD, a co-senior author of the study and professor of physiology at UCSF. “We were pleased to find the drug was tremendously successful in normalizing neuronal and cognitive function with lasting effects.”
TBI is a leading cause of long-term neurological disability, with impairments in concentration and memory affecting patients’ quality of life. It’s also the strongest environmental risk factor for dementia – even a mild concussion significantly increases an individual’s risk.
Reversing the Effects of Concussion
ISRIB was developed in the lab of co-senior author Peter Walter, PhD, who was a professor of biochemistry at UCSF at the time of the study, now emeritus and at Altos Labs. Previous studies demonstrated that the molecule could effectively block the integrated stress response and showed evidence of improving cognition and behavior across different mouse models of TBI. However, the cellular mechanisms by which ISR inhibition restored cognition remained unknown.
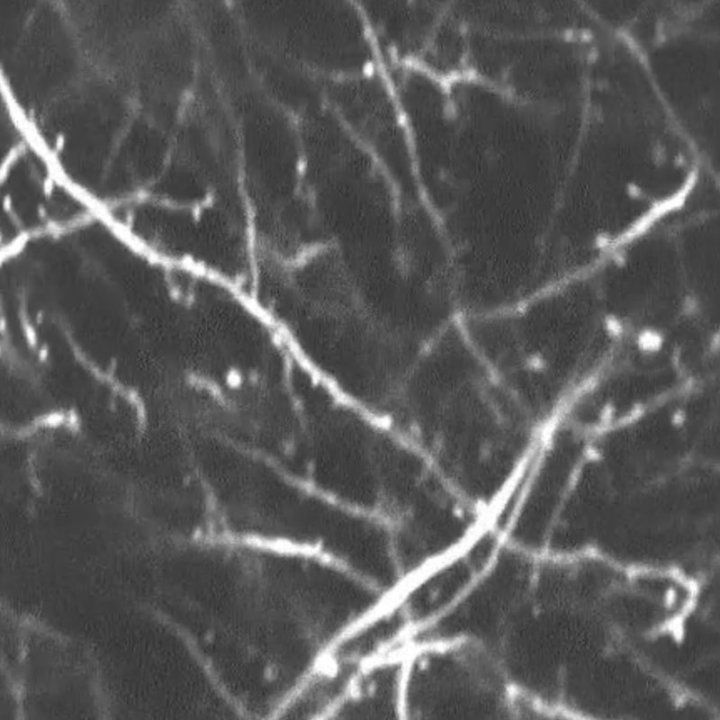
To investigate this, UCSF graduate student Elma Frias, PhD, began to explore how the ISR and its inhibition affected neurons in the parietal cortex, a brain region involved in working memory.
Using advanced imaging techniques, Frias observed the effects of TBI on dendritic spines, the primary site of excitatory communication between neurons, over the course of multiple days.
In healthy conditions, neurons show a fairly consistent rate of spine formation, maturation and elimination – dynamics that support learning and memory. But after a single mild concussion, mouse cortical neurons showed a massive burst of newly formed spines and continued to make excessive spines for as long as they were measured.
“Some may find this counterintuitive at first, assuming more dendritic spines would be a good thing for making new memories,” said co-senior author Susanna Rosi, PhD, a professor of physical therapy and neurological surgery at UCSF at the time of the study, now also at Altos Labs. “But in actuality, having all too many new spines is like being in a noisy room – when too many people are talking, you can’t hear the information you need.”
The brain is very plastic; it can be rewired and healed.
These new spines didn’t stick around for very long, however, and most were removed within days, meaning they hadn’t formed lasting functional synaptic connections.
These aberrant dynamics were rapidly reversed once mice were treated with ISRIB. By blocking the ISR, the drug was able to repair the neuronal structural changes resulting from the brain injury and restore normal rates of spine dynamics. These neuronal structural alterations were also associated with an improvement in performance to normal levels in a behavioral assay of working memory, which persisted for over a month after the final treatment.
“A month in a mouse is several years in a human, so to be able to reverse the effects of concussion in such a lasting way is really exciting,” said Frias.
Restoring the Brain’s Potential to Heal
The authors suggest that TBI triggers a persistent activation of the ISR, which in turn leads to the ongoing proliferation of transient spines that fail to support memory formation. Future experiments will explore whether ISRIB has similar effects on other cell types, brain areas and cognitive tasks.
ISR activation has been implicated in many neurological disorders, including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis (MS) and amyotrophic lateral sclerosis (ALS). Thus, the researchers believe ISRIB may have therapeutic potential in multiple patient populations.
While there was no evidence of the drug’s toxicity in mice, clinical trials are currently evaluating the safety and effectiveness of ISRIB in humans.
“This study reminds us that the brain is very plastic; it can be rewired and healed,” said Rosi. “By briefly inhibiting this stress pathway, we may be able to restore healthy synaptic and cognitive function in many neurological conditions.”
Disclosures: Peter Walter has a patent application for the invention of ISRIB, and rights to the invention have been licensed by UCSF to Calico.
Authors: Co-authors of this study include Mahmood S. Hoseini, Karen Krukowski, Maria Serena Paladini, Katherine Grue and Kira D.A. Rienecker, all at UCSF, as well as Gonzalo Ureta at Fundación Ciencia & Vida in Santiago, Chile.
Funding: This study was funded, in part, NIH grants R01AG056770 R01EY002874, R01MH122478 and R25GM056847, NSF grant 1822650, and the generous support of the Roger’s family and the Weill Innovation Award.