For many infectious diseases, a strategy for drug design is clear: Find something that kills what's causing the disease that won't kill the patient. But what's the best approach when our own cells spread within and make us sick - as when normal cells turn cancerous?
Drugs that target a disease-causing bacterium or fungus often block the activity of a protein that pathogenic cells need to survive. But most proteins in cancer cells also are present in normal cells. That makes it difficult to target tumor cells without causing harmful side effects in healthy tissue.
The question has no definitive answer. But a new key to fighting cancer might be found in a cell's skeleton or, more precisely, its cytoskeleton, says Thomas Huckaba, PhD. Cytoskeletons in normal cells often differ from those within cancer cells.
The cell's "bones" comprise a network of filaments. But unlike the humans in whom they reside, cells can be shape shifters. To make these shape changes possible, the cytoskeleton changes shape in turn - filaments lengthen and contract.
A cell needs filaments to maintain or change shape, to divide and to transport cargo between different parts within the cell. The largest of these filaments, called microtubules, are made up entirely of the protein tubulin.
Skeletons in the Cancer Cell Closet
 |
Thomas Huckaba. |
Huckaba, a postdoctoral fellow at UCSF in the laboratory of Ronald Vale, PhD, believes that a telltale modification of tubulin might be used to distinguish cancer cells from normal cells. If that's true, and if it's possible to track down the source of this modification, it might also be possible to one day use this knowledge to better target cancer cells for destruction.
Cells can not only change shape due to microtubule changes, but also can move across the landscape of bodily tissues. We can be thankful that this occurs during normal development and during wound healing. But scientists view metastasis, the process by which a cancer spreads from its initial site to other parts of the body, as cell migration gone wrong.
Scientists have observed tubulin modifications when cells migrate. The modifications occur specifically at the edge of a cell closest to the direction of migration. These tubulin changes during cell migration are the focus of Huckaba's research.
Tubulin can exist in a variety of forms, Huckaba explains. To give rise to these forms, an array of different enzymes "label" tubulin with chemical modifications at specific sites along the protein. In many cases, scientists remain in the dark about both the purpose of the modifications and the identity of the modifying enzymes. "We've known about these modifications for so long, but nobody's been able to identify the enzymes," Huckaba says.
Tracking Down an Elusive Enzyme
Huckaba hopes to learn the purpose of a specific kind of tubulin modification called acetylation. For reasons unknown, this acetylation appears to accumulate in cells in some cancers. Cancerous cells within certain types of breast and colon tumors have much higher levels of acetylated tubulin than normal breast and colon cells, for instance.
Huckaba wants to know how tubulin acetylation changes microtubules and the cells containing these modified microtubules. Huckaba believes that tubulin acetylation may play a role both in normal cell migration and in metastasis of certain cancer types.
To study this tubulin modification, Huckaba first needs to identify the responsible enzyme. "Maybe there's a link between acetylation and cancer," he says. "But until we actually have the enzyme, it's going to be hard to tell."
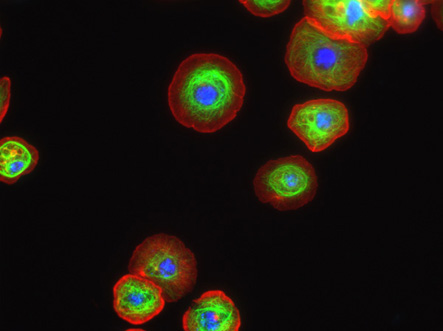 |
Humans have bones; cells have cytoskeletons. In these cells a major component of the cytoskeleton, called microtubules, are stained green. Another component of the cytoskeleton, the actin filaments, are stained red. DNA in the nucleus is stained blue. UCSF Postdoctoral fellow Thomas Huckaba works with fruit fly cells to investigate biochemical alterations in microtubules that may play a role in human cancer. |
Huckaba is taking a few different approaches to find this mysterious enzyme. First, he is screening a collection of cells with mutations. He is looking for cells that do not contain acetylated tubulin. The bereft cells are likely to contain mutations in genes that encode the proteins involved in acetylation. Huckaba is also using a second strategy to identify the enzyme, an approach he calls "bucket biochemistry." He will separate all the proteins in an extract prepared from cells through a series of purification steps. He will track tubulin-acetylating activity, until he is left with a single purified protein capable of performing this modification in a test tube.
The Road to Cancer Treatments
Eventually, Huckaba hopes that the discovery of this acetylating enzyme could provide clues to certain types of cancer. "Because we know that there are increases in tubulin modifications in certain cancer types," he says, "knowing the enzyme responsible and how it is regulated will help in the development of diagnostics."
If there is an important link between tubulin acetylation and cancer - as the high levels of acetylation in cancer cells suggest - then the measurement of the acetylating enzyme in cells from biopsy samples could become an important tool to identify certain cancer types.
Huckaba hopes this research will go beyond diagnostics. "We may be able to target cancer drugs to specifically knock this enzyme down," he says. "But that's five or six steps down the road at this point."
Huckaba's studies of tubulin acetylation are funded through a fellowship from the Damon Runyon Cancer Research Foundation.
Photo/Thomas Huckaba
Related Links:
Vale Lab

