Newly discovered genetic disease sheds light on body's water balance
Two infant boys whose bodies were overloaded with excess fluid have led UCSF pediatricians to the discovery of a new genetic disease. In the process, they have discovered a rare type of mutation where different substitutions in a single amino acid cause two different, opposite genetic disorders.
The new disorder, called Nephrogenic Syndrome of Inappropriate Antidiuresis (NSIAD), is described in the May 5 issue of The New England Journal of Medicine.
“This discovery gives better insights into treating these patients and potentially many others,” said Stephen Gitelman, MD, principal author of the study and professor of clinical pediatrics at the University of California, San Francisco. “It sheds new light on the mechanisms that the body uses to maintain fluid homeostasis—the correct balance of fluids needed for health and life.”
Gitelman and a team of fellow pediatrician scientists at UCSF took their findings about the two patients to the laboratory, working with colleagues to isolate the genetic mutations responsible for their disruption in water balance.
They found that each infant has a different mutation in a specific gene, AVPR2, that encodes the V2 receptor (V2R) for vasopressin, a hormone that instructs the kidneys to retain water. Neither patient was producing measurable levels of vasopressin, yet the V2 receptor on cells in the collecting duct of the kidneys remained activated as if it was binding to the hormone.
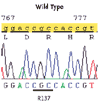
Both mutations activate the receptor by changing a single amino acid, arginine, located on the gene at position 137. This location, R137, already is known to scientists who study fluid homeostasis. A third, different mutation had been previously shown to block the V2 receptor, causing a condition opposite to NSIAD called Nephrogenic Diabetes Insipidus (NDI). In that condition, instead of retaining fluids, the kidneys excrete water excessively, leading to severe dehydration.
“To our knowledge, this is the only reported example in which mutations affecting the same amino acid cause two different genetic diseases,” the UCSF researchers write.
While only two infants so far have been identified with NSIAD, the condition may not be rare, according to Stephen Rosenthal, MD, professor of pediatric endocrinology and co-lead author of the study with endocrinology fellow Brian Feldman, MD, PhD. “Water retention is a common problem, and with new tools we can examine our long-held assumptions about its cause,” Rosenthal said. “There may be mutations in other components of the V2 receptor signaling cascade that result in inappropriate antidiuresis.”
In fact, after Feldman presented this work at an international meeting last September, he said, “Several physicians indicated they were now considering the diagnosis of NSIAD as an explanation for low sodium levels in their pediatric patients.”
MEDICAL MYSTERY
The discovery of NSIAD began in 2003 after two unrelated baby boys, each about three months old, were referred to UCSF Children’s Hospital with symptoms of fluid overload and strikingly low levels of sodium in their blood. The first child suffered from severe irritability. A few months later, the second infant was admitted with generalized seizures.
Doctors in the pediatric endocrinology and pediatric nephrology services at UCSF Children’s Hospital determined that each baby’s kidneys were inappropriately retaining water, and the excess fluids were diluting blood sodium levels.
The usual diagnosis for this condition is Syndrome of Inappropriate Antidiuretic Hormone Secretion (SIADH)—a common medical problem whose cause was discovered almost 50 years ago to be an overabundance of vasopressin (also called antidiuretic hormone). However, when the UCSF doctors took advantage of new, rapid tests to measure vasopressin in these two infants, they found no detectable levels of the hormone in either child’s bloodstream. They concluded that some other mechanism was disrupting fluid homeostasis.
“This is a good example of the value of humility about what we know and don’t know in clinical medicine,” said Gitelman. “When something seems to fit a classic pattern, but actually doesn’t, our job as physician scientists is to recognize the discrepancy and get to the bottom of the situation.”
First, Gitelman and his colleagues worked to find an innovative treatment for the two young patients. “When adults retain too much water we can treat them by restricting fluid intake,” he said. “We can’t do that with infants—almost all of their nutrition comes in liquid form.”
The physicians were reluctant to use other medications that generate increased water loss in the kidneys, as these can have side effects or toxicities in young children. Instead, with Feldman and pediatric endocrinology fellow Eric Huang, MD, Gitelman administered urea, a natural nitrogen compound that encourages urination by acting as an osmotic agent. As water flowed out of their bodies and sodium concentrations returned to normal, both babies recovered. They continue to be followed by the UCSF team.
Next Gitelman, Rosenthal and Feldman took blood samples from each baby boy and his mother to search for the cause of the unique disorder. They learned that Raymond Fenwick, a scientist with Quest Diagnostics Nichols Institute, was developing an assay that could be used to sequence the AVPR2 gene that codes for the V2 receptor.
Fenwick sequenced the patients’ AVPR2 genes and found that each boy had a unique mutation affecting the amino acid arginine at position 137 (R137). The single nucleotide change resulted in one baby having a change from arginine to cysteine (R137C). This same mutation was carried by his mother. The other child had a change from arginine to leucine (R137L) but the mother is normal, suggesting that the baby’s mutation occurred spontaneously. The fact that the two affected patients are boys is not a coincidence, as the AVPR2 gene is X-linked and only inherited from the mother’s side, but not the father’s side of a family.
The scientists teamed up with Gabriel Vargas, MD, PhD, a UCSF assistant adjunct professor of psychiatry conducting research in the laboratory of Mark von Zastrow, PhD, on how cell surface receptors carry messages into the cell that activate specific events. The V2 receptor has been widely studied as an example of a large family of proteins called G protein-coupled receptors (GPCR), which are involved in signal transduction and are responsible for many physiological processes.
Normally, when vasopressin binds to the V2R, it stimulates a chain of events inside the cell. Vesicles containing the water channel Aquaporin 2 (AQP-2) are shuttled from the cytoplasm to the apical membrane of the collecting duct kidney cells. This process increases water permeability and diminishes urine output from the kidneys.
The group set out to prove that changing the amino acid at position 137, from arginine to either cysteine or leucine, caused the AVPR2 to produce a mutated V2 receptor that is always “on,” inappropriately signaling the kidneys to retain water. The opposite condition, diabetes insipidus, is caused when the arginine at R137 is changed to histidine, and the resulting V2 receptor signals the kidneys to excrete water excessively.
To test the hypothesis that V2 receptors were active in their patients even though vasopressin was absent, Feldman developed an assay for receptor activity. One marker of this activity is an increase in the amount of cyclic AMP (cAMP) within the cell. He worked in the laboratory of pediatric endocrinologist Walter Miller, MD, using a method to determine cAMP levels in cells by using a luciferase plasmid that responds to cAMP.
Luciferase is the substance that lights up in fireflies. Feldman showed that wild-type cells contain low levels of light from luciferase—and thus low levels of cAMP.
In Feldman’s experiments, cells with the R137C mutation proved to have four times as much cAMP as wild-type cells; those with the R137L mutation have 7.5 times as much. With no trigger from the vasopressin hormone, the V2 receptors nevertheless were active.
Feldman noted that an interesting aspect of the study was the time that it required, in comparison to the painstaking efforts required to isolate genes a decade ago. In nine months, this collaborative team was able to go from recognizing subtle new symptoms to identifying a new genetic disease. Feldman said, “This experimental research is an example of how close the basic science laboratory bench has moved to the patient’s bedside in modern medicine. These two areas now complement each other and bench-to-bedside progress leads to more rapid advances that benefit our patients.”
The authors note in the New England Journal article, “The V2 receptor has been an important tool for understanding the physiology of water balance—(Also) it has served as a prototype for G protein-coupled receptor biology. Further characterization of NSIAD may offer additional insights into fluid homeostasis and clinical disease, as well as expand our understanding of GPCR signaling.”
The group is now conducting a series of further studies in both the clinic and the laboratory to better understand how these mutations affect water balance.
This work was supported in part by NIH training grants to researchers Feldman and Huang and by an NIH grant to the UCSF Pediatric Clinical Research Center.
NOTE REGARDING AUTHORS: Brian J. Feldman, MD, PhD, and Stephen M. Rosenthal, MD, share the lead authorship of this study. Stephen E. Gitelman, MD, is principal author. Additional authors are Gabriel A. Vargas, MD, PhD, Eric A. Huang, MD, Mina Matsuda-Abedini, MD, Robert H. Lustig, MD, Robert S. Mathias, MD, Anthony A. Portale, MD, and Walter L. Miller, MD, all of UCSF. Co-author Raymond G. Fenwick, PhD, is employed by Quest Diagnostics, Inc. and developed the clinical assay used for sequencing the AVPR2 gene in this study.
ILLUSTRATION: A schematic diagram published with the NEJM article shows how results differ when the amino acid arginine at position 137 is substituted by cysteine or leucine (causing NSIAD) or substituted by histidine (causing diabetes insipidus). Permission to publish this diagram may be requested from Karen Pedersen at The New England Journal of Medicine, (781) 434-7847, [email protected].
COMMENTARY: A commentary appears on page 1847 of the May 5 New England Journal of Medicine: “Hyperactive Vasopressin Receptors and Disturbed Water Homeostasis” by Nine V.A.M. Knoers, MD, PhD, of Radboud University, the Netherlands.