New UCSF Mission Bay Center to Study Membrane Proteins
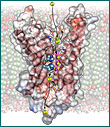
An atomic-scale image of a functioning ammonia channel in cell membranes, determined by the Stroud lab and published last year in Science.
(See larger view)
Membrane proteins connect the worlds inside and outside the cell, and although they are critical for all life functions, they are poorly understood because they are difficult to extract from the fatty membrane in fully functional form - an essential step in order to purify them and study their shape and function. Membrane proteins serve as "gatekeepers," controlling movement of molecules into or out of cells. They can also report the presence of important extra-cellular signals, and they anchor cells in their correct place. Membrane proteins make up about 30 percent of all proteins in higher organisms. Research at the new Membrane Protein Expression Center will be carried out at Mission Bay campus sites, where most of the principal investigators direct their laboratories, and also at UCLA and the University of Nebraska. Center scientists develop and apply new ways to express and purify many classes of membrane proteins in amounts sufficient to study their structure and function. One goal is to obtain three-dimensional atomic structures of the channels and transporter proteins that regulate movement of material between cells and their environment. Other classes of membrane proteins act as transducers for hormonal signals, neuronal signaling in the brain and as channels in cardiovascular function. Availability of the proteins in functional form will allow researchers to determine the molecular cell biology and drug interactions of many therapeutically and biologically important membrane proteins. This is expected to have a major impact on efforts to understand the basic biochemistry behind transmembrane signaling and transport mechanisms. It will also have an enormous impact on drug discovery aimed at cell surface receptors. "So far, there has been virtually no success in expressing human or even mammalian membrane proteins," said Robert Stroud, director of the new center and a QB3 faculty affiliate at UCSF."We intend to make these proteins available to the community in pure and functional form, in ways useful for analysis of their function, and for drug discovery. The center is transforming the biology of membrane proteins." Stroud, a professor of biochemistry and biophysics and also of pharmaceutical chemistry at UCSF, is a highly regarded X-ray crystallographer whose laboratory has determined in unprecedented detail the structure of key channel proteins, including the recent determination of the atomic-level structure of the channel that controls movement of ammonia into and out of cells. The structure was featured on the cover of Science, which published the findings. The new center is funded by the Sandler New Technologies Fund and by the National Institute of General Medical Sciences, as part of the NIH "Roadmap Initiative" aimed at overcoming critical roadblocks to research essential to human health. Source: Wallace Ravven