Can a New Drug Candidate Cure Pancreatic Cancer?
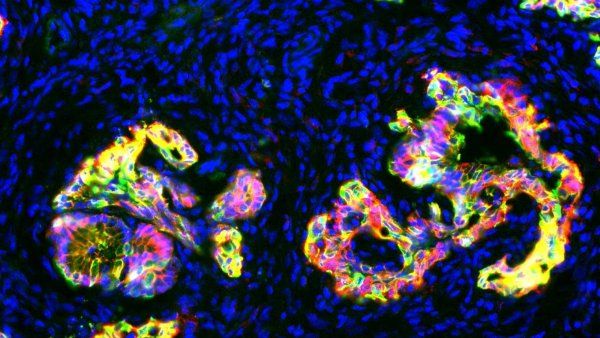
A new drug candidate permanently modifies a wily cancer-causing mutation, paving the way for making pancreatic cancer treatable, or perhaps even curable.
University of California San Francisco
Give to UCSFA new drug candidate permanently modifies a wily cancer-causing mutation, paving the way for making pancreatic cancer treatable, or perhaps even curable.

Delivering medicine through amniotic fluid is as effective as delivering it to the fetal brain via cerebrospinal fluid to treat serious disordrs such as Angelman syndrome.

UCSF scientists found a way to predict Alzheimer’s disease up to seven years before symptoms appear by analyzing patient records with machine learning. Conditions that most influenced prediction of Alzheimer’s were high cholesterol and, for women, osteoporosis.

Thirty-two UCSF scientists are among the most influential individuals in their respective fields, according to the most recent analysis of research citations by the science and intellectual property company, Clarivate.

UCSF researchers are working across disease specialties. Diabetes researchers are looking at how oncologists use CAR T-cell therapy to reprogram a person’s immune system to attack cancer cells, for example. They hope to similarly reprogram the immune system to fight diabetes.

Engineered immune cells. Supercharged scans. Drug implants. Gene manipulators. Blood biopsies. Read how these breakthroughs are transforming cancer care.

Two UCSF scientists – James Gardner, MD, PhD, and Rebeca de Pavia Fróes Rocha, PhD – have received Pew awards for their work in immunology as part of a program that supports promising early-career investigators.

Oncology specialists from around the globe will gather for the American Society of Clinical Oncology (ASCO) Annual Meeting to discuss the latest cancer therapies, technologies, research and education.

UCSF researchers discover how gabapentin interacts with calcium channels, opening doors for more effective treatments in epilepsy and Lupus by influencing channel biogenesis.
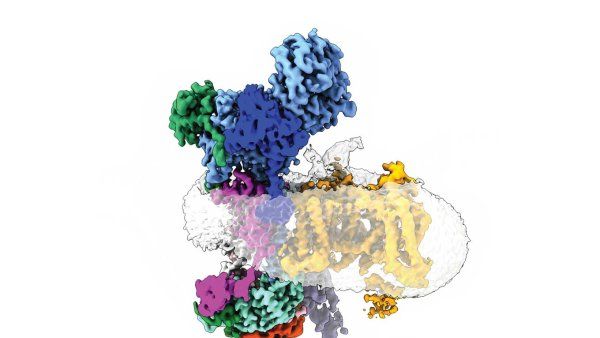
A traditional African psychedelic plant medicine called ibogaine is the blueprint for two new drug candidates that could treat addiction and depression.
Henry Bourne, MD, a long-time UCSF researcher who chronicled its rise to prominence in the 1970s, has died at the age of 83.

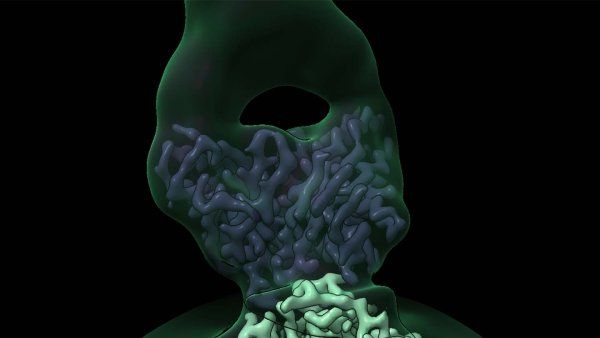
In a first, UCSF scientists created a molecular-level, 3D picture of how an odor molecule activates a human odorant receptor.

Angela Phillips, PhD, leads research that could help predict future viruses like COVID and the antibodies we might use to treat them.
